 PDF(2377 KB)
PDF(2377 KB)


Seasonal dynamic on biomass and morphological structure of absorptive root between female and male Fraxinus mandshurica
JIA Hongtao, WEI Xing, LI Wei, WU Chunze, WEI Qingyu, HAN Chunli
Journal of Nanjing Forestry University (Natural Sciences Edition) ›› 2026, Vol. 50 ›› Issue (2) : 149-157.
 PDF(2377 KB)
PDF(2377 KB)
 PDF(2377 KB)
PDF(2377 KB)
Seasonal dynamic on biomass and morphological structure of absorptive root between female and male Fraxinus mandshurica
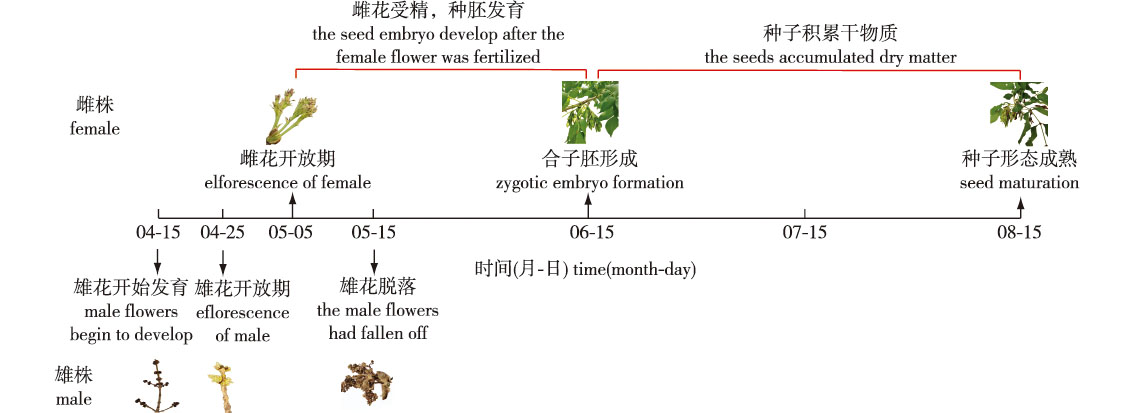
【Objective】This study aims to clarify the relationship between absorptive root development and reproductive processes in dioecious Fraxinus mandshurica, investigate gender dimorphism in root traits, and provide insights for sex-specific nutrient management strategies.【Method】Using mature female and male F. mandshurica trees, fine root samples (orders 1-3) were collected across the growing season (April-August) via soil coring. Root biomass, morphology, anatomical structure, and mycorrhizal infection rate were analyzed using root scanning and paraffin sectioning techniques.【Result】Absorptive root biomass increased seasonally in both sexes. Before male flowering, root biomass was similar between genders. From male flowering to female flowering, male root biomass increased by 33.14%, while female biomass remained unchanged. During seed development, both sexes exhibited marked increases in root biomass-females by 481.63% and males by 393.90% (P<0.05).Root length density was consistently higher in males, with the 1st-order root length density being 2.27 times greater in males at seed maturity. Gender-specific seasonal patterns were observed in root diameter, with male 1st-order roots being 21.80% (June) and 15.52% (August) thicker than females (P<0.05). Anatomical analysis revealed that this was due to greater cortical thickness in males (11.57% and 7.73% higher in June and August, respectively; P<0.05), while vascular bundle diameter did not differ. Mycorrhizal infection rate was also higher in males (by 4.45% in June and 3.84% in August).【Conclusion】Seasonal dynamics of absorptive root biomass in F. mandshurica are closely linked to reproductive phenology. Males exhibited earlier root development and maintained higher absorptive root biomass throughout the growing season. The observed gender differences in root morphology, anatomy and mycorrhizal association highlight distinct belowground strategies between female and male trees.
Fraxinus mandshurica / dioecy / absorptive root / root biomass / root morphology / root anatomy / seasonal dynamics
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
谷加存, 王东男, 夏秀雪, 等. 功能划分方法在树木细根生物量研究中的应用:进展与评述[J]. 植物生态学报, 2016, 40(12):1344-1351.
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
杜欣竹. 不同发育阶段水曲柳菌根和根毛的时空异质性[D]. 哈尔滨: 东北林业大学, 2018.
|
| [19] |
盛萍萍, 刘润进, 李敏. 丛枝菌根观察与侵染率测定方法的比较[J]. 菌物学报, 2011, 30(4):519-525.
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
卫星, 刘颖, 陈海波. 黄波罗不同根序的解剖结构及其功能异质性[J]. 植物生态学报, 2008, 32(6):1238-1247.
|
| [44] |
|
| [45] |
王文娜, 王燕, 王韶仲, 等. 氮有效性增加对细根解剖、形态特征和菌根侵染的影响[J]. 应用生态学报, 2016, 27(4):1294-1302.
|
/
| 〈 |
|
〉 |