 PDF(2815 KB)
PDF(2815 KB)


Establishment of a protoplast isolation system for Ziziphus jujuba ‘Dongzao’ fruit
GUO Yanmeng, HU Dechun, ZHANG Shulin, YOU Yang, ZHU Gaopu, TIAN Li, ZHOU Ruijin, DONG Wenyi, ZHANG Mengmeng
Journal of Nanjing Forestry University (Natural Sciences Edition) ›› 2026, Vol. 50 ›› Issue (2) : 78-84.
 PDF(2815 KB)
PDF(2815 KB)
 PDF(2815 KB)
PDF(2815 KB)
Establishment of a protoplast isolation system for Ziziphus jujuba ‘Dongzao’ fruit
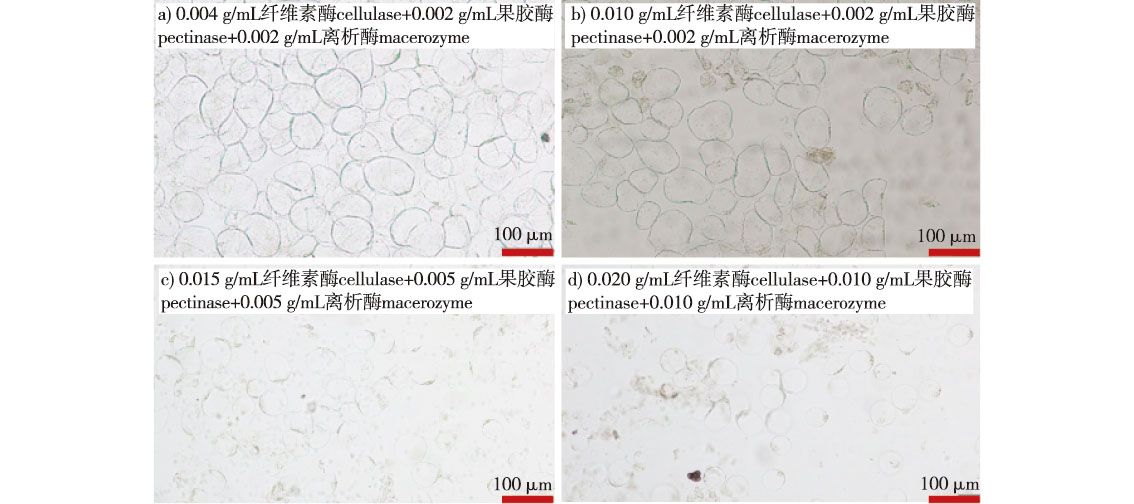
【Objective】This study aims to establish an efficient protoplast isolation system for Ziziphus jujuba ‘Dongzao’ fruit. By systematically analyzing the effects of various factors on protoplast extraction, it seeks to provide technical support for subsequent research on gene function and the molecular mechanisms underlying fruit development.【Method】Using semi-red fruits of Z. jujuba ‘Dongzao’ as experimental material, a multi-factor experimental design was applied to optimize key parameters. The factors investigated included: enzyme combination (cellulase, macerozyme, pectinase), KCl concentration (150, 205, 255, 305, 355, 405, 430, 455, 505 mmol/L), enzymatic hydrolysis duration (2.0, 2.5, 3.0, 3.5, 4.0 h), and vacuum (200 Pa) infiltration time (10, 20, 30 min). Through systematic screening, the optimal concentrations of cellulase, pectinase, macerozyme, and KCl were determined, leading to the identification of the most effective enzyme solution for protoplast isolation.【Result】The optimal enzymatic solution consisted of 0.02 g/mL cellulase, 0.01 g/mL pectinase, 0.01 g/mL macerozyme, 430 mmol/L KCl, 65 mmol/L CaCl2, 10 mmol/L MES (2-morpholinoethanesulfonic acid), and 1 mmol/L DTT (dithiothreitol), adjusted to pH 5.8. The protoplast yield reached 1.82 × 106 /mL when the tissues were subjected to vacuum infiltration at 200 Pa for 20 min, followed by 3.0 h of enzymatic digestion in the dark on a horizontal shaker. Under these conditions, the isolated protoplasts exhibited intact morphology and high viability.【Conclusion】By screening four key factors—enzyme concentration, hydrolysis time, KCl concentration, and vacuum treatment duration—an efficient enzymatic protocol was established for isolating protoplasts with intact cellular structure and high viability from Z. jujuba ‘Dongzao’ fruit. This study successfully develops a robust protoplast separation system, which provides a reliable technical foundation for future genetic functional studies and the molecular analysis of fruit development in Z. jujuba ‘Dongzao’.
winter jujube (Ziziphus jujuba ‘Dongzao’) / fruit / protoplast / enzymatic method / enzymatic solution
| [1] |
|
| [2] |
彭邵锋, 陆佳, 陈永忠, 等. 木本植物原生质体培养体系研究进展[J]. 中国农学通报, 2013, 29(1):1-6.
|
| [3] |
何珊珊, 李宏宇, 马月, 等. 百合原生质体分离培养和瞬时转化[J]. 浙江大学学报(农业与生命科学版), 2025, 51(1):67-79.
|
| [4] |
李妮娜, 丁林云, 张志远, 等. 棉花叶肉原生质体分离及目标基因瞬时表达体系的建立[J]. 作物学报, 2014, 40(2):231-239.
|
| [5] |
杨冬, 周静, 段青, 等. 大丽花花瓣高效的原生质体分离体系研究[J]. 江西农业学报, 2024, 36(8):70-78.
|
| [6] |
张学英, 葛会波, 刘艳萌, 等. 草莓原生质体分离条件的研究[J]. 分子植物育种, 2006, 4(增刊2):147-152.
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
赵维峰, 孙光明. 毛叶枣与冬枣原生质体分离体系的建立[J]. 热带作物学报, 2006, 27(3):51-55.
|
| [18] |
朱道圩, 秦永华, 郅玉宝, 等. 软枣猕猴桃原生质体培养与细胞团再生的初步研究[J]. 河南农业大学学报, 2001, 35(3):221-224.
|
| [19] |
王玖瑞, 刘孟军, 代丽. 枣树组织培养研究进展[J]. 果树学报, 2002,(5):336-339.
|
| [20] |
王志慧, 杨新平, 茆军, 等. 外源菌剂对棉花秸秆堆肥过程中微生物群落及腐熟效果的影响[J]. 生态与农村环境学报, 2024, 40(10):1376-1384.
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
霍信屹, 陈欣晨, 赵慧敏, 等. 郁金香叶肉原生质体的分离纯化与瞬时转化方法[J]. 浙江大学学报(农业与生命科学版), 2025, 51(1):80-88.
|
| [26] |
|
/
| 〈 |
|
〉 |